How it works
A simple journey, from profile to pre-screening
AccessTrial structures information, speeds up trial sorting and improves routing to the right investigators.
Build your profile
Gather and upload the essential medical elements: medical reports, test results, genetic abnormalities, ongoing treatments
Launch AI-assisted matching
The platform analyses your file and identifies compatible trials.
Submit your application
Identify the investigator centres best suited to your situation and submit your application online.
Why AccessTrial
A medical report is all it takes
Check eligibility from a medical report less than one month old and get the list of suitable clinical trials in under 5 minutes.
No Data Restructuring Required
Our advanced natural language processing works directly with unstructured patient summaries. Just upload or copy paste your latest medical report.
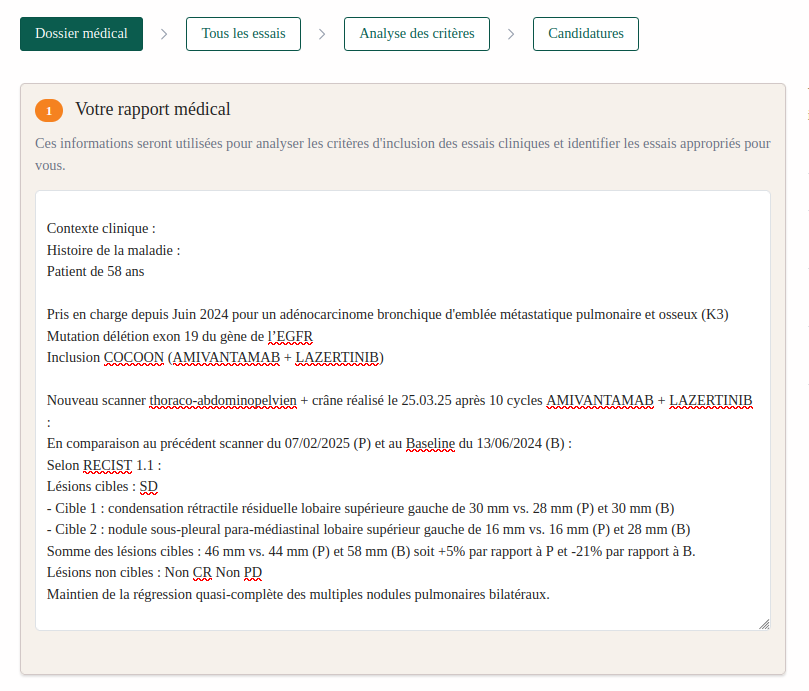
Criterion-by-Criterion Analysis
Each eligibility criterion is evaluated individually, with detailed explanations, highlighting compatibility areas and points to verify before submitting the application.
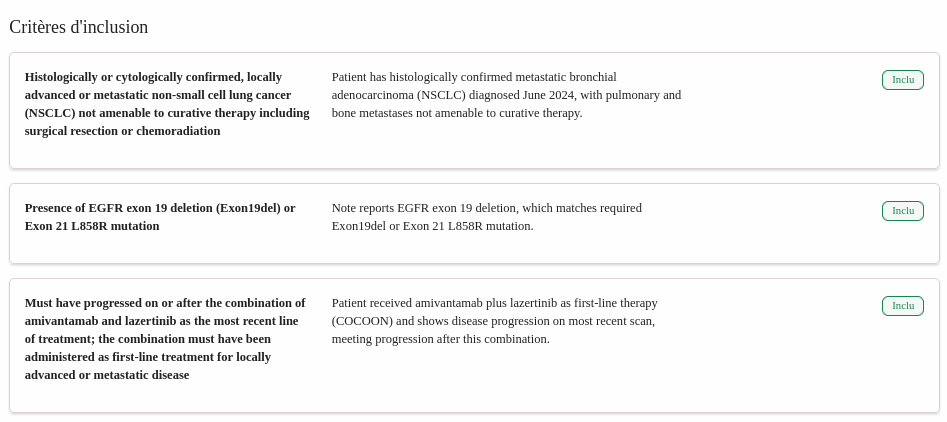
Proven Clinical Accuracy
Validated on thousands of patient records with expert-level accuracy, assisting medical professionals and reducing pre-screening time by 42%.
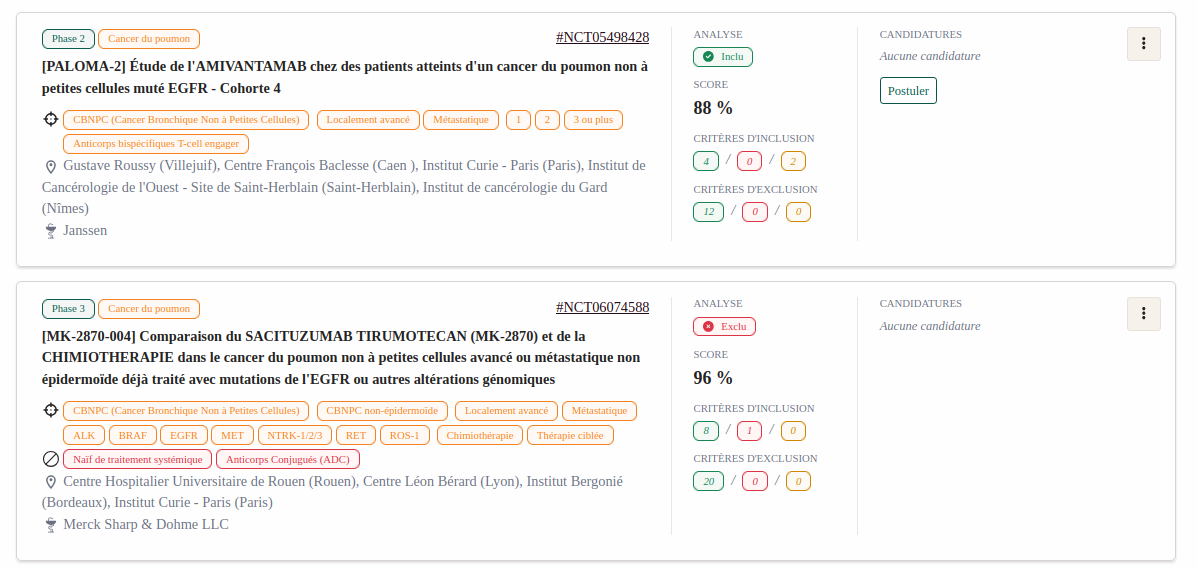
Access to innovation
By facilitating access to clinical trials, AccessTrial offers new perspectives to patients by proposing innovative therapeutic options.
Medical data security
Medical data is hosted in France on HDS (Health Data Host) infrastructure and processed in strict compliance with GDPR.
Time saving
By automating the pre-screening process, AccessTrial enables doctors and patients to save precious time in identifying clinical trials.
User journeys
A user journey tailored to your profile
Understand, qualify and refer.
Users
What users say
We have been waiting for such a tool for many years!
Pr Mahasti Saghatchian
Onco-Senologist, Head of the Oncology department
Hôpital Américain
AccessTrial multiplies and accelerates the capacity for research and access to clinical trials in France.
Dr Stéphane Champiat
Medical Oncologist, Department of Innovation and Early Trials
MD Anderson
AccessTrial makes it possible to target open trials in a specific clinical situation.
Pr Nicolas Girard
Onco-pulmonologist, Head of the Medical Oncology department
Institut Curie Montsouris
Finding the most suitable clinical trial for our patients is a daily challenge.
Pr Elise Deluche
Onco-Senologist, Head of department
CHU Limoges
Care cannot be conceived without the help of clinical research and the best treatment.
Pr Stéphane Oudard
Medical Oncologist, Head of department
Hôpital Européen Georges Pompidou





